Table 1 from Absolute standard hydrogen electrode potential measured by reduction of aqueous nanodrops in the gas phase. | Semantic Scholar
![PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/56964684a624c5af38c7e62256db3faa4c542d88/19-Table2-1.png)
PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar

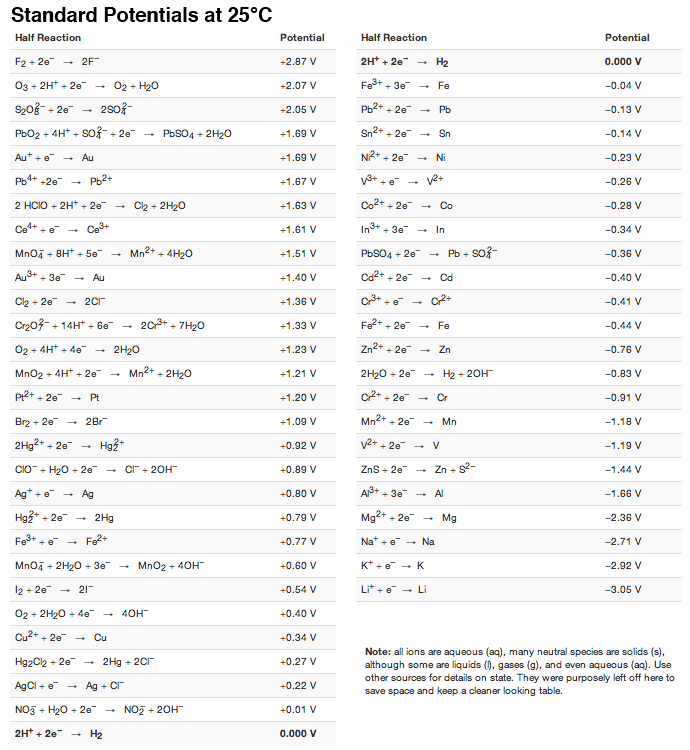
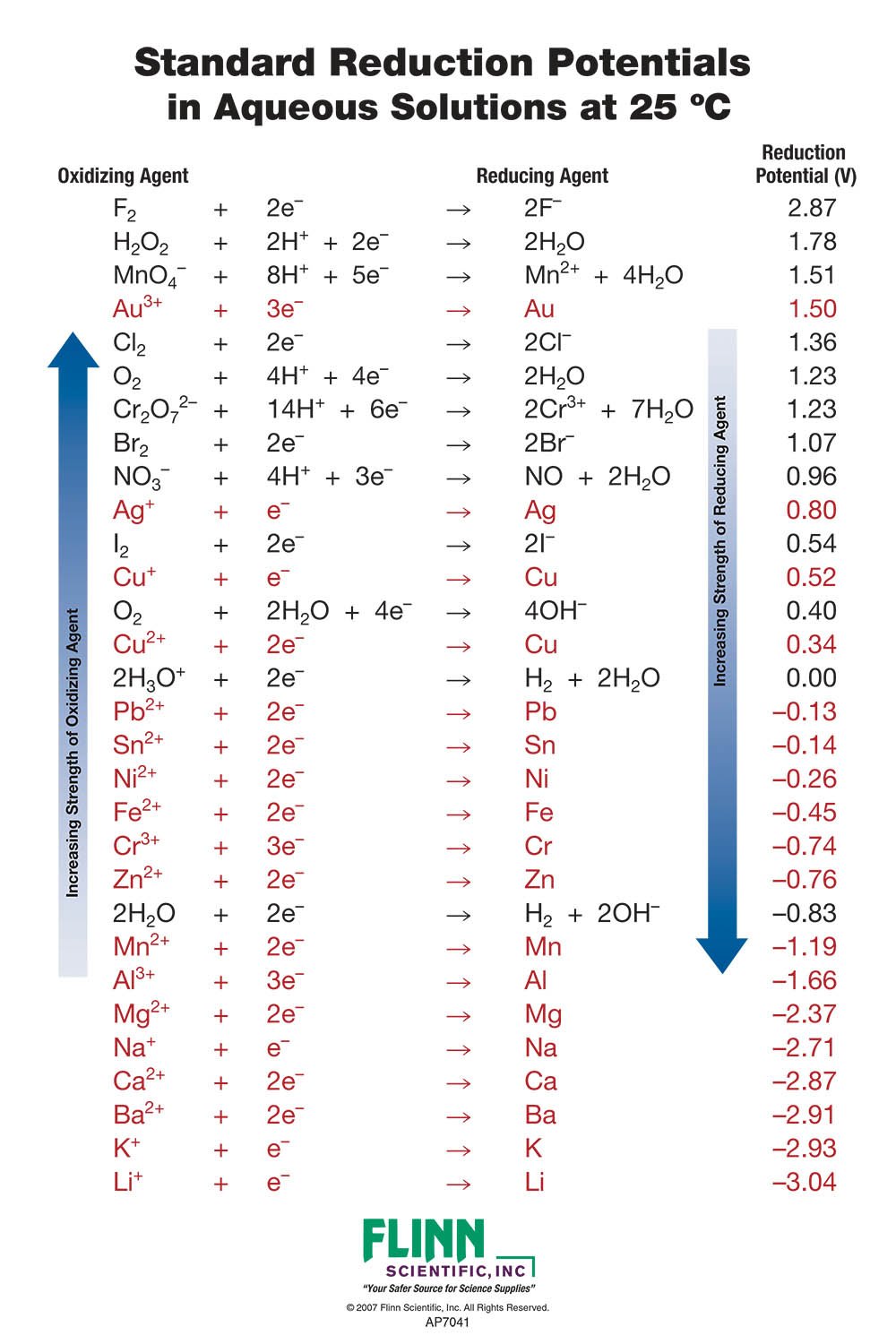
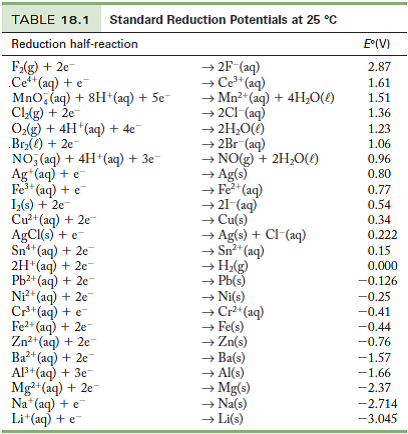
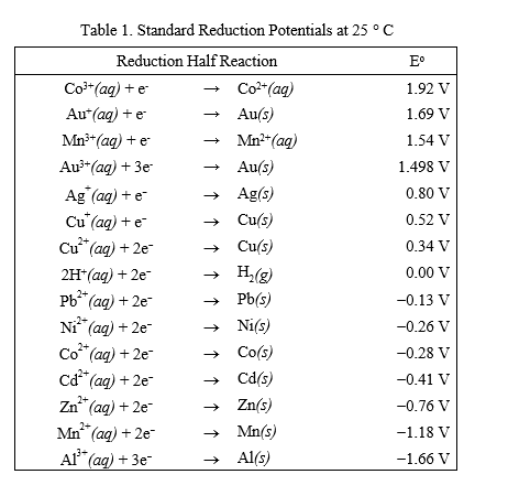
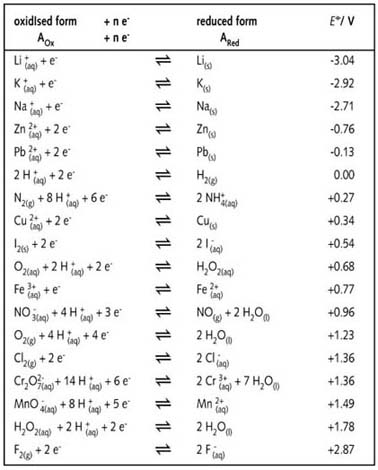
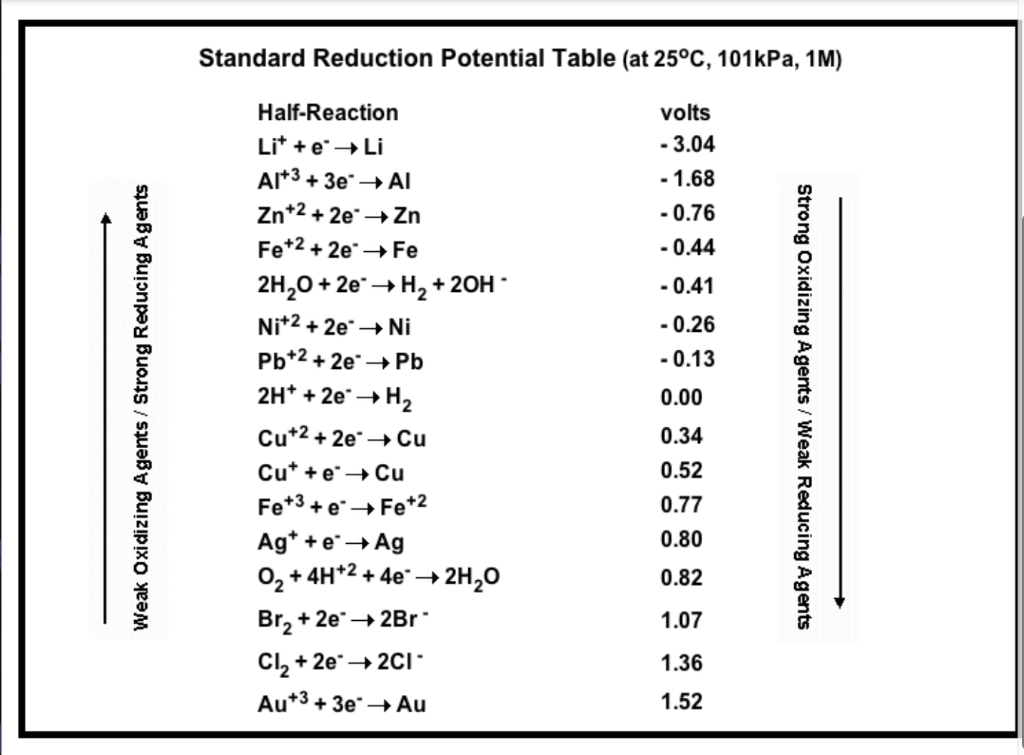
Consult the table of standard electrode potentials and suggest three substances that can oxidise ferrous ions under suitable conditions

Using the standard electrode potentials given in the Table, predict if the reaction between the following is feasible. Ag(s) and Fe^3 + (aq)

Using the standard electrode potentials given in the table, predict if the reaction between the following is possible. Ag^+(aq) and Cu(s)





:max_bytes(150000):strip_icc()/Standardreductionpotential-5b551731c9e77c003ec223b3.jpg)